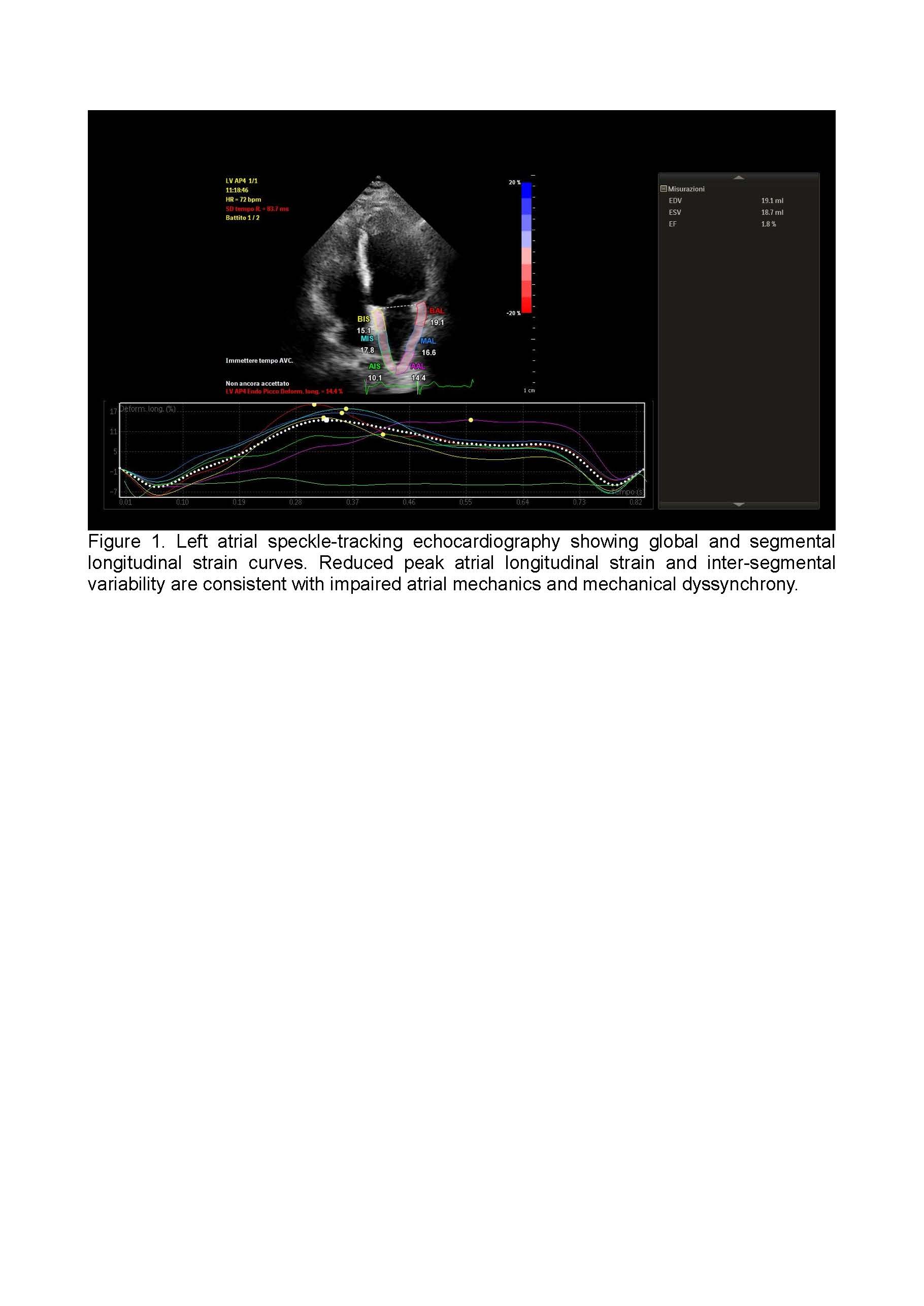
Background: Atrial fibrillation (AF) is a multifactorial arrhythmia in which epicardial adipose tissue (EAT) has emerged as a key contributor to atrial structural, electrical, and functional remodeling. Left atrial epicardial adipose tissue (LA-EAT), through endocrine, paracrine, and mechanical mechanisms, promotes inflammation, fibrosis, and atrial dyssynchrony. Early identification of atrial dysfunction is essential to improve risk stratification and guide therapeutic strategies. Methods: We performed a narrative review focusing on the pathophysiological relationship between left atrial epicardial adipose tissue and atrial remodeling in patients with atrial fibrillation. Particular attention was given to non-invasive imaging techniques, with emphasis on speckle-tracking echocardiography for the assessment of left atrial strain. Strain parameters, including reservoir, conduit, and contractile function, were analyzed in relation to epicardial adipose tissue burden and markers of atrial remodeling. In addition, emerging therapeutic strategies targeting epicardial adipose tissue were reviewed for their potential impact on atrial function and arrhythmia outcomes. Results: Reduced left atrial strain parameters are consistently associated with increased LA-EAT and advanced atrial remodeling in patients with atrial fibrillation. Strain imaging allows early detection of atrial dysfunction before overt structural changes occur and improves identification of patients at higher risk of AF recurrence. The integration of strain analysis with volumetric atrial assessment enhances diagnostic accuracy and patient stratification. Therapeutic interventions such as weight reduction and GLP-1 receptor agonists show promising effects in reducing EAT volume and mitigating atrial remodeling, potentially improving ablation outcomes. Conclusions: Left atrial strain imaging represents a valuable non-invasive tool for assessing EAT-related atrial remodeling in patients with atrial fibrillation. Its integration into routine clinical practice may optimize patient selection, guide therapeutic decisions, and improve long-term outcomes in AF management.