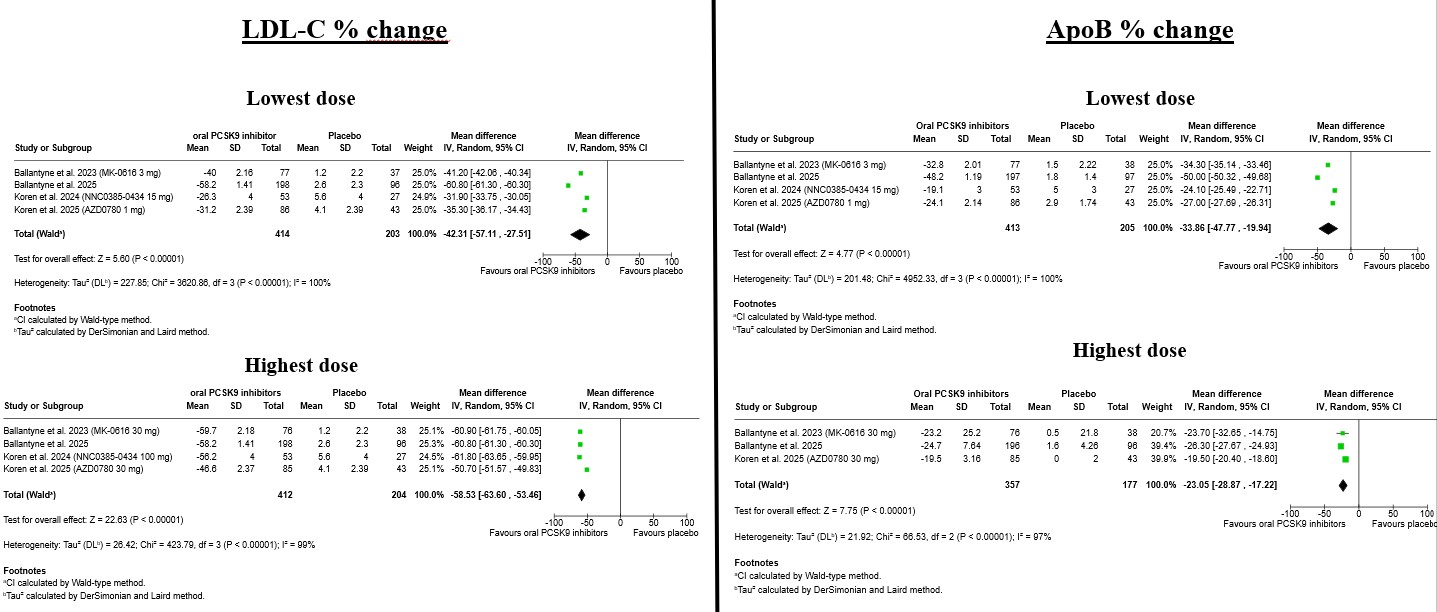
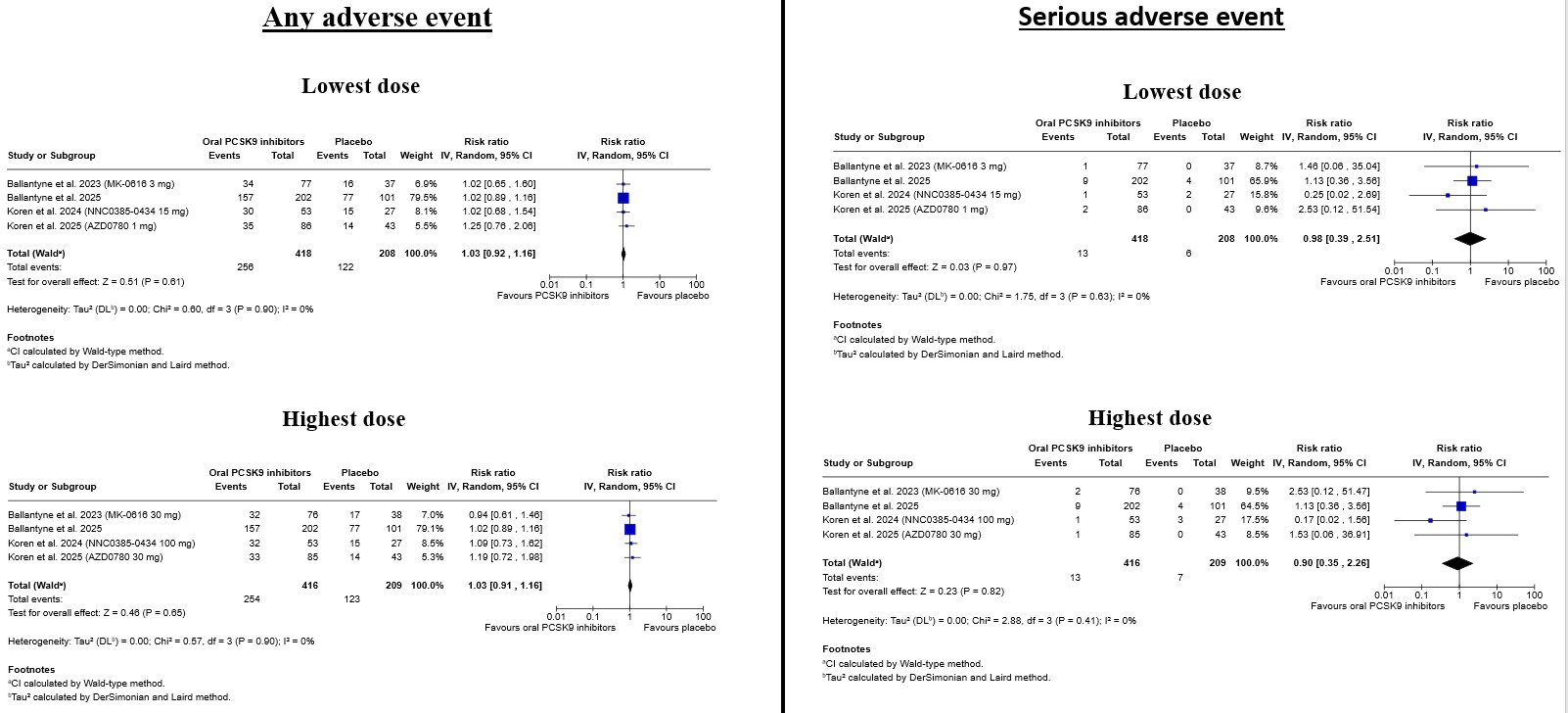
Background: Oral inhibitors of proprotein convertase subtilisin/kexin type 9 (PCSK9) represent a novel lipid-lowering strategy that may overcome limitations related to injectable therapies. However, the dose-specific efficacy and safety of this emerging drug class have not been comprehensively evaluated. Methods: We performed a systematic review and dose-stratified meta-analysis of randomized, placebo-controlled trials evaluating oral PCSK9 inhibitors. PubMed, Embase, and CENTRAL were searched from inception to November 2025. Primary efficacy outcomes were percentage changes in LDL cholesterol (LDL-C) at the lowest and highest tested doses. Secondary endpoints included apolipoprotein B (ApoB), non–HDL cholesterol, and lipoprotein(a) [Lp(a)]. Safety outcomes comprised overall adverse events (AEs), serious AEs, and selected individual AEs. Random-effects models were applied, and interaction testing was used to assess dose–response relationships. Results: Four trials encompassing 859 participants were included. Oral PCSK9 inhibitors significantly reduced LDL-C at both dose levels, with greater reductions observed at the highest dose (-58.5%) compared with the lowest dose (-42.3%; p for interaction = 0.02). Similar dose-dependent effects were observed for ApoB (-49.7% vs -33.9%) and non–HDL cholesterol, while reductions in Lp(a) were consistent but not significantly dose-dependent. Safety outcomes were comparable to placebo across doses, with no increase in overall or serious AEs. Rates of gastrointestinal, musculoskeletal, and infectious events were low, and no dose-related safety signals were identified. Conclusions: Oral PCSK9 inhibitors provide substantial, dose-dependent reductions in atherogenic lipoproteins with a favorable short-term safety profile. These findings support continued clinical development and highlight the potential role of oral PCSK9 inhibition as an effective and patient-friendly option for intensive LDL-C lowering.