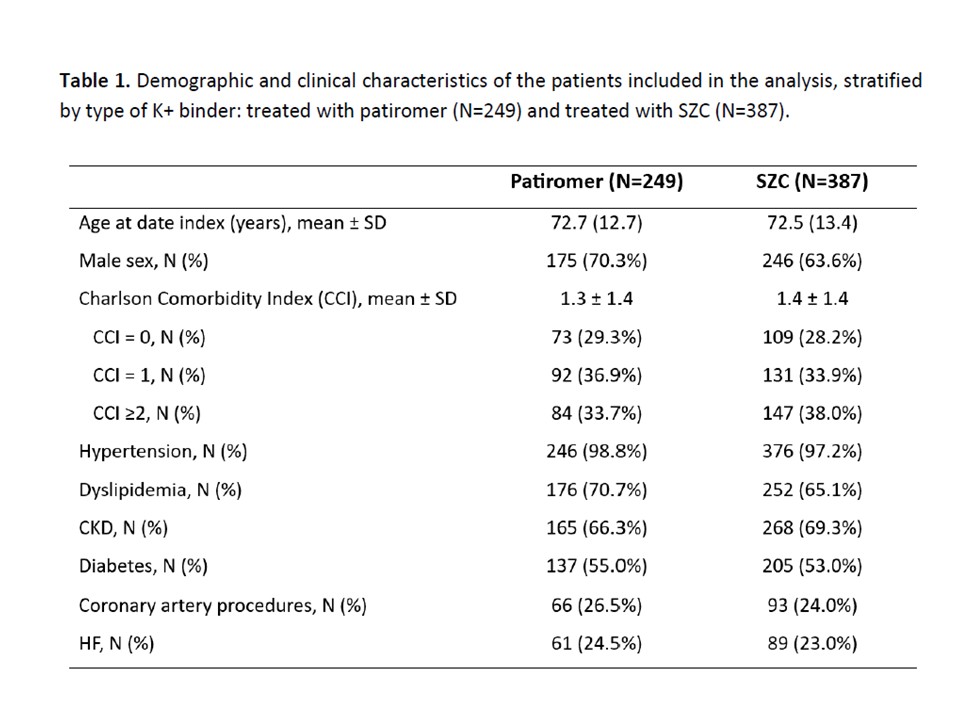
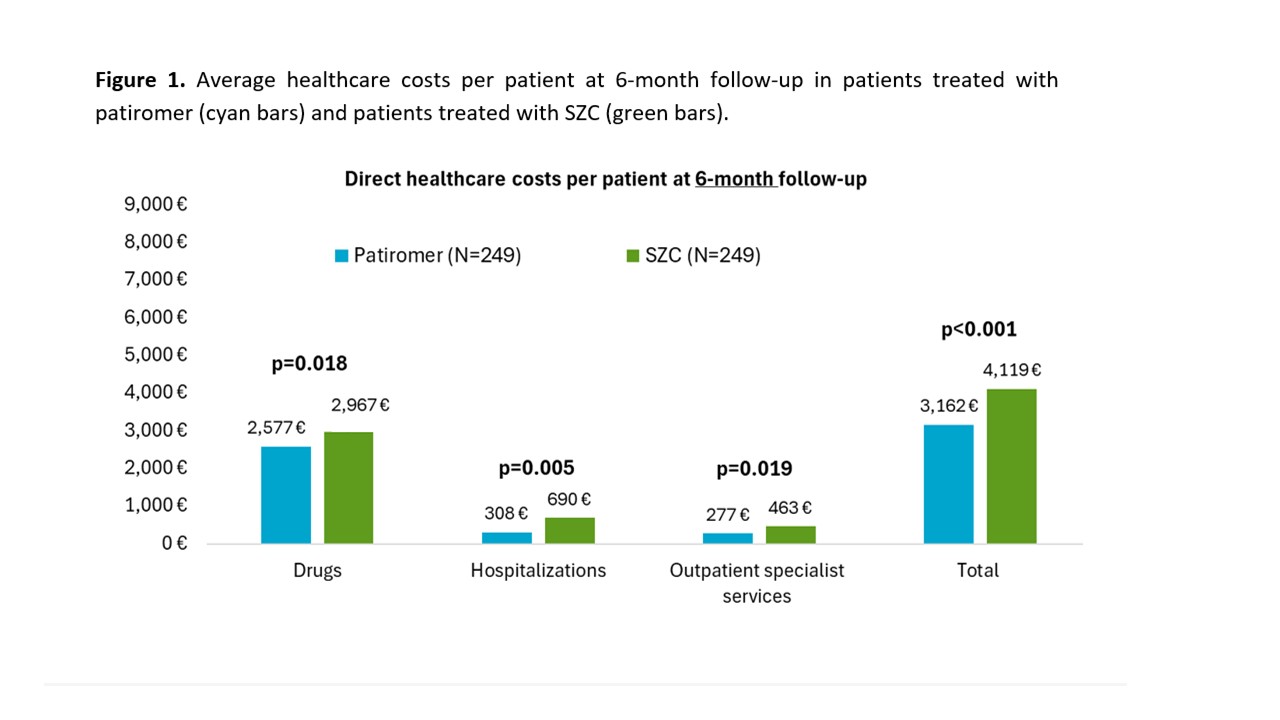
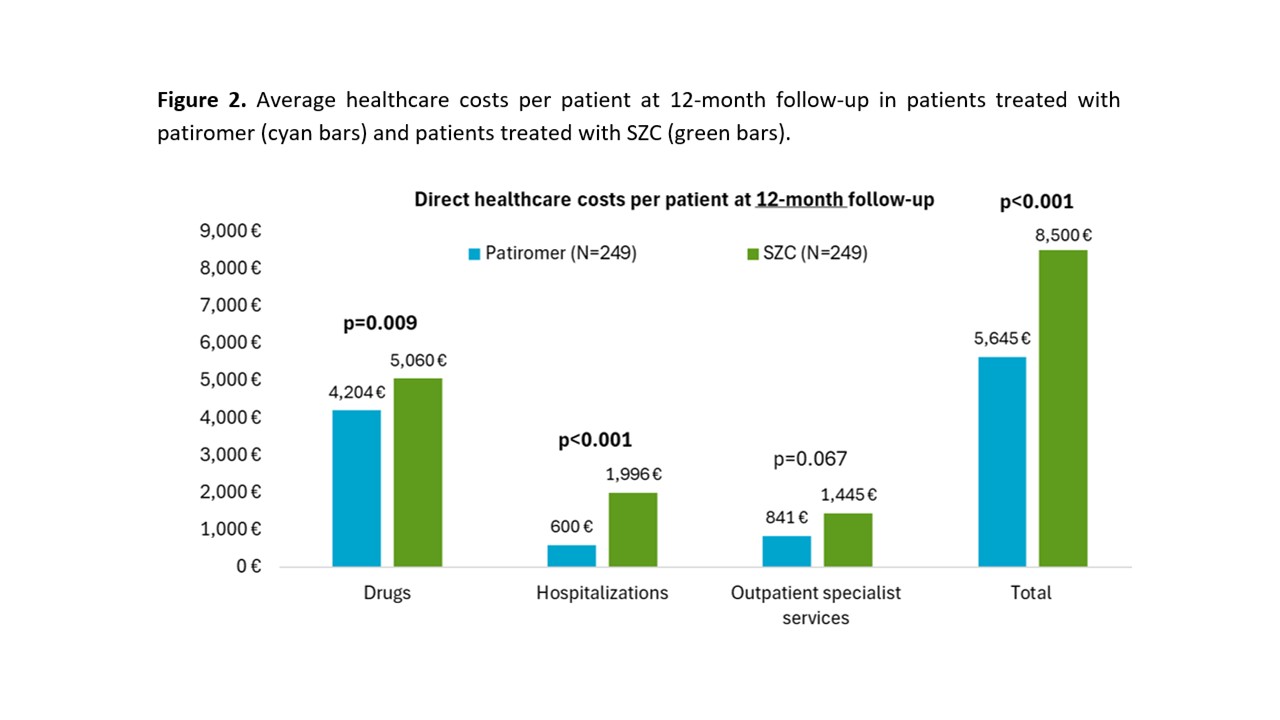
Hyperkalemia is common in patients with chronic kidney disease (CKD) and heart failure (HF), often affecting prognosis and treatment continuity with renin–angiotensin–aldosterone system inhibitors (RAASi). Potassium (K⁺) binders allow control of K + serum levels and maintenance of RAASi therapy at proper dosage. Given the scarcity of real-world data in Italy, this analysis focused on patients treated with different K⁺ binders, assessing the consistency with data from the US setting, where outcome differences in favor of non–sodium-exchange binders have emerged. From administrative databases of Italian healthcare bodies (~6 million beneficiaries), adults with ≥1 prescription of patiromer or sodium zirconium cyclosilicate (SZC) between Jan-2022 and Jun-2024 were selected. Patients on dialysis, transplant recipients, those treated with ≥1 K⁺ binder or with <6 months of follow-up from the first K⁺ binder prescription were excluded. Demographic and clinical characteristics, treatment adherence at 6 months—estimated using the Medication Possession Ratio (MPR: treatment duration/observation time)—and costs were described. Clinical outcomes were assessed after matching (by age, sex, comorbidities, and co-medications) at 6 months and over the entire observation period (10.7 months for patiromer; 7.5 months for SZC). Among the 636 included patients treated with patiromer (N=249) or SZC (N=387), the mean age was nearly 73 years, with male predominance. The most common comorbidities were hypertension, hyperlipidemia, CKD, diabetes, and HF (Table 1). During the first 6 months of follow-up, the proportion of adherent patients (MPR ≥80%) was significantly higher in patiromer than in SZC users (67.5% vs 20.4%; p<0.001), a finding confirmed over the entire period (42.5% vs 13.4%; p<0.001). After matching, the rate of HF-related hospitalizations was lower with patiromer compared with SZC, both at 6 months (5.4 vs 12.8 events per 100 patient-years) and over the entire follow-up (5.3 vs 15.7). Mean per-patient costs were lower with patiromer at 6 months (€3,162 vs €4,119) and at 12 months (€5,645 vs €8,500), mainly due to reduced hospitalization costs (Figures 1-2). Considering pharmaceutical expenses, patiromer users had lower costs for anti-anemic and antidiabetic drugs. In Italian clinical practice, patiromer treatment led to reduced HF hospitalization risk and healthcare costs compared with SZC, supporting its use in hyperkalemia management for patients with CKD and HF.