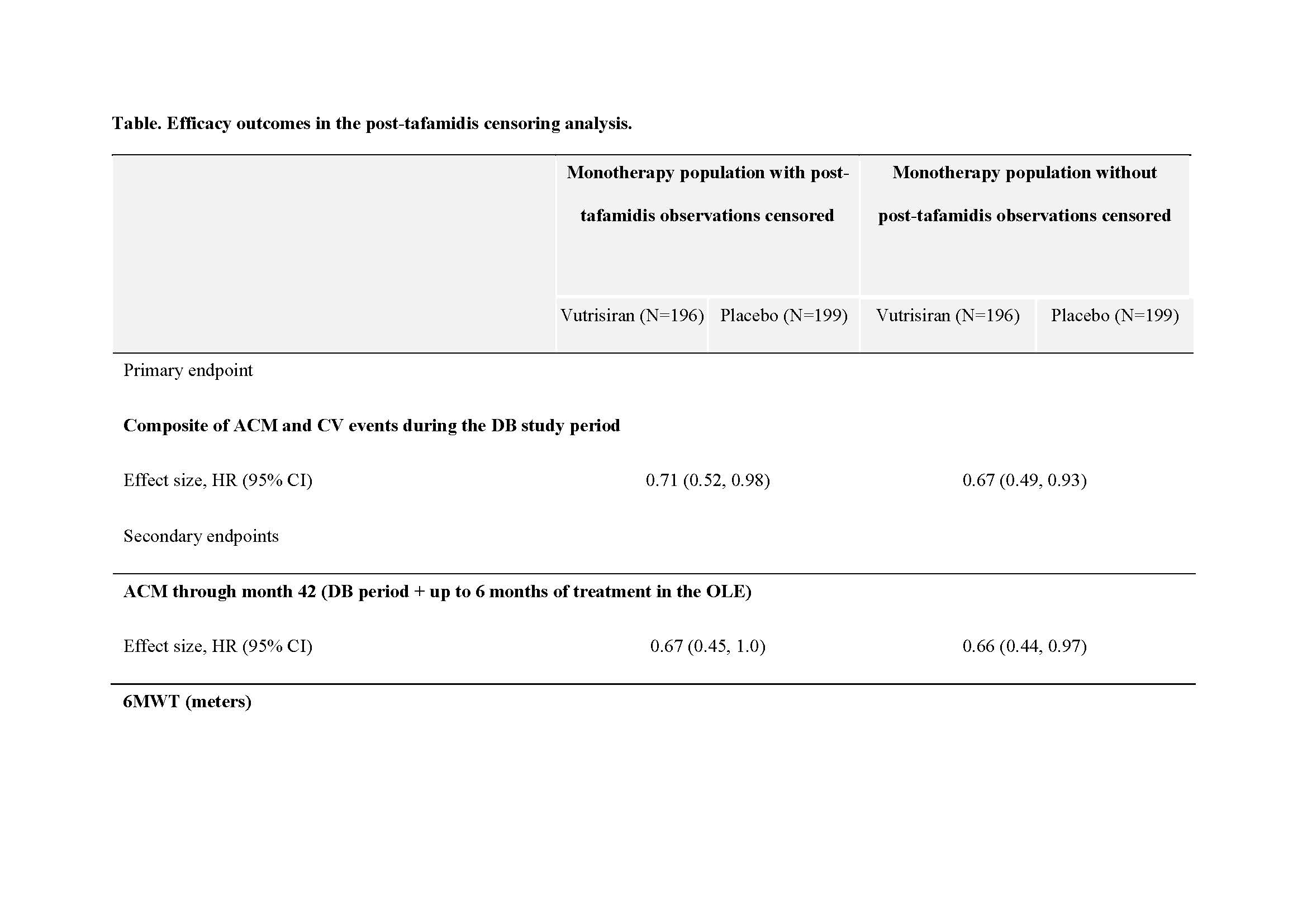
Introduction: In HELIOS-B(NCT04153149), vutrisiran significantly reduced the risk of the primary endpoint composite of all-cause mortality(ACM) and recurrent cardiovascular(CV) events, and met all secondary endpoints(including ACM at 42months), vs placebo in patients with transthyretin amyloidosis with cardiomyopathy(ATTR-CM) in both the overall and monotherapy (patients not receiving tafamidis at baseline) populations. In the monotherapy population, 21.5% of patients initiated tafamidis after randomization during the double-blind period. Hypothesis: Consistent results with the primary analysis are expected when observations after tafamidis initiation are censored in the monotherapy population of HELIOS-B(H-B). Methods: Patients were randomized 1:1 to receive vutrisiran 25mg or placebo every 12weeks for up to 36months. Patients who were not receiving tafamidis at baseline (monotherapy population) could initiate tafamidis after enrollment depending on the investigator's decision and the availability of tafamidis. This analysis censored patient observations in the monotherapy population post-tafamidis initiation and then repeated the primary analyses of the H-B study. Results: A total of 395 patients in H-B were in the vutrisiran monotherapy population and were included in this analysis. Of these, 85(21.5%) initiated tafamidis post randomization(vutrisiran n=44; placebo n=41) during the double-blind period. The results from this post hoc analysis are consistent with the results of the original primary analysis of the monotherapy population, and demonstrate the efficacy of vutrisiran vs placebo, when observations post-tafamidis initiation were censored. The hazard ratios for vutrisiran vs placebo were 0.67 and 0.71 for the composite primary endpoint of ACM and recurrent CV events and 0.66 and 0.67 for the secondary endpoint of ACM before and after censoring, respectively. The results were also consistent for other secondary endpoints ( Table ). Conclusion: The efficacy of vutrisiran vs placebo is consistent after the observations following tafamidis initiation in the monotherapy population of H-B were censored. This provides further evidence supporting the efficacy of vutrisiran as monotherapy in patients with ATTR-CM. Funding: This study was funded by Alnylam Pharmaceuticals.