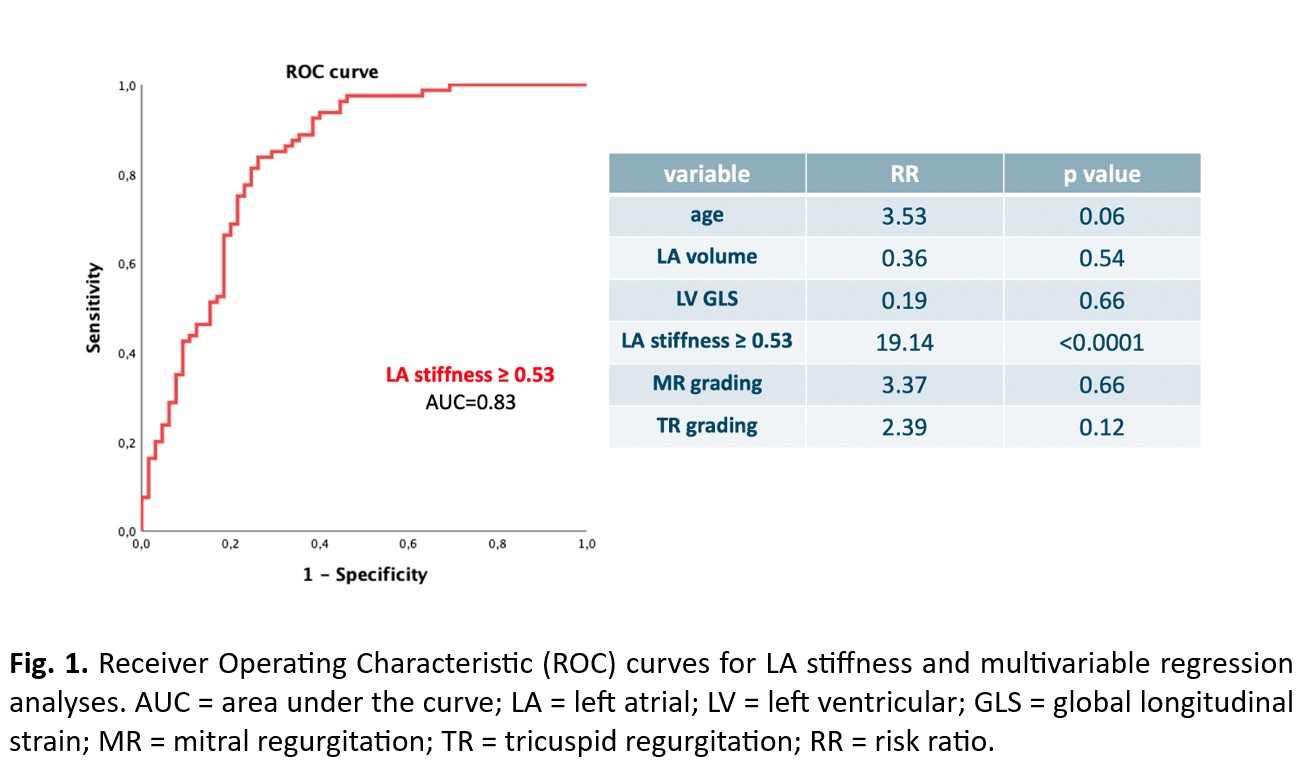
Background. Pulmonary hypertension (PH) has a complex diagnostic pathway. Differential diagnosis between left heart disease (LHD) (group 2) and lung diseases (group 3) PH is also challenging. A comprehensive echocardiographic evaluation for suspected PH is recommended to assess cardiac etiology. The aim of this study was to assess the potential value of speckle tracking echocardiography (STE), as a sensitive marker of myocardial performance, to predict LHD in patients presenting with dyspnea and PH. Methods. Consecutive outpatients with dyspnea on exertion and subsequent diagnosis of PH were retrospectively enrolled. Inclusion criteria were NYHA class ≥ II, left ventricular (LV) ejection fraction ≥ 50%, echocardiographic evidence of tricuspid regurgitation (TR) velocity > 2.8 ms, confirmed diagnosis of PH with relative etiology (heart failure with preserved ejection fraction (HFpEF) or lung disease). Patients underwent clinical, biohumoral and advanced echocardiographic evaluation. Primary endpoint was the prediction of HFpEF. Results. Overall, 145 patients were enrolled (80 with HFpEF, 65 with lung disease). Mean age was 75±12 years, 53% were female. Patients with HFpEF were older (77±10 vs. 68±14 years, p < 0.0001) and had higher LA volume (p < 0.0001), E/e’ (p < 0.0001). Regarding STE parameters, patients with HFpEF had lower left atrial reservoir strain (LASr) (15±8 vs. 24±11%, p < 0.0001) and worse LV global longitudinal strain (GLS) (-13±9 vs. -17 ± 6%, p = 0.009) compared to patients with lung diseases. LA stiffness, an index of LA dysfunction and fibrosis calculated as the ratio between E/e’ and LASr, was higher in patients with HFpEF (median LA stiffness = 1.09 [confidence interval (CI) = 0.64 – 1.73] vs 0.35 [CI = 0.20 – 0.59], p < 0.0001). With receiver operating characteristic curves, both E/e’ and LASr provided a good prediction for HFpEF (AUC 0.73 and 0.79 respectively), but LA stiffness significantly enhanced the predictive power (AUC = 0.83) with an optimal cutoff value ≥ 0.53. With multivariate analysis including age, LA volume, LV GLS, LA stiffness, mitral and TR grade, LA stiffness ≥ 0.53 (RR = 19.14, [CI 5.86 – 62.55]) was the only independent predictor of HFpEF in our cohort of patients with PH (Fig.1). Conclusions. STE may aid differential diagnosis of etiology between LHD and lung diseases in patients with PH. The combination of E/e’ and LASr to calculate LA stiffness offers the most accurate prediction of PH with cardiac etiology.