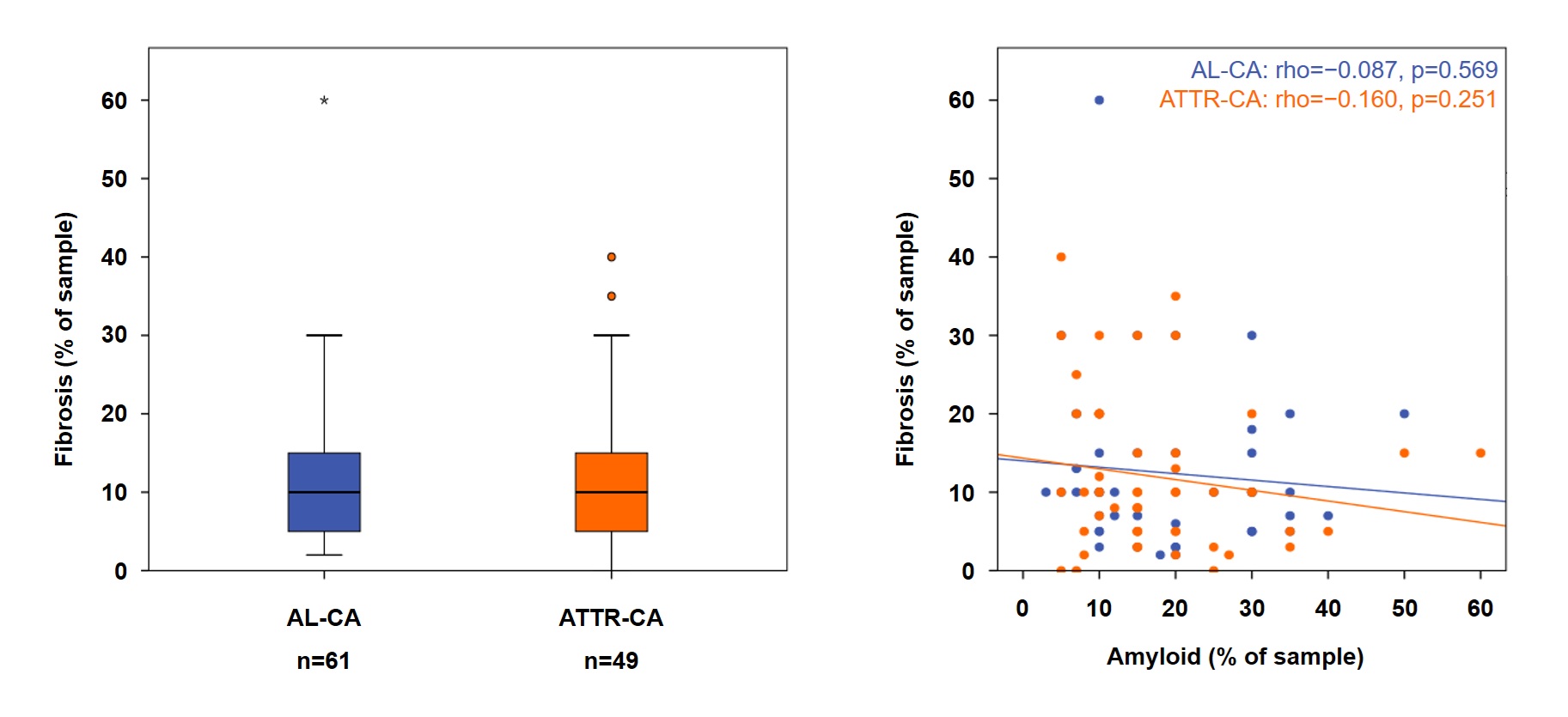
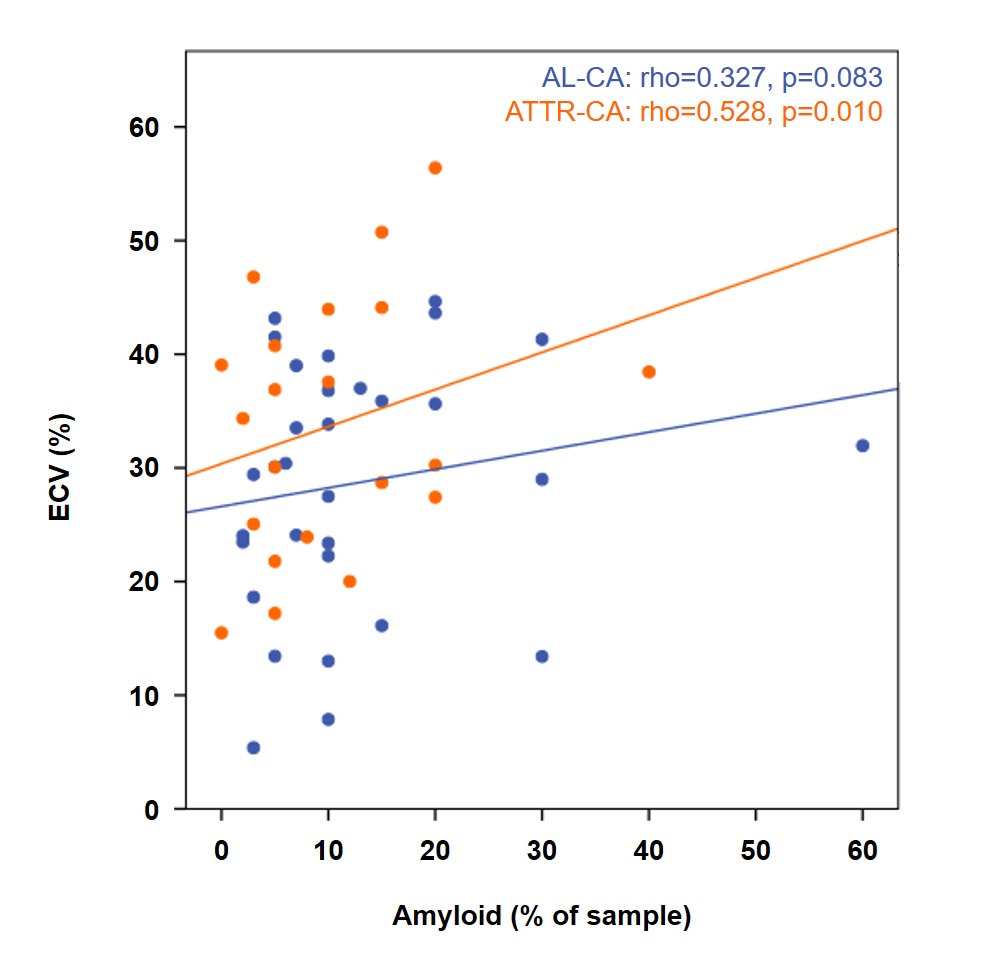
Background: Treatment options for cardiac amyloidosis (CA) are rapidly expanding. Therapies suppressing further amyloid accumulation may enable partial amyloid reabsorption; emerging monoclonal antibodies may further enhance amyloid clearance. Myocardial fibrosis, triggered by cardiac injury and amyloid deposition, may be less reversible. Fibrosis data in CA remain limited and mostly derived from explanted hearts. Methods: We analyzed consecutive patients undergoing left ventricular (LV) endomyocardial biopsy for suspected CA at a tertiary referral center; patients with prior myocardial infarction were excluded. Histology quantified fibrosis and amyloid burden and measured inflammatory cell density. Cardiovascular magnetic resonance (CMR) was performed as clinically indicated and extracellular volume (ECV) was assessed. Results: The cohort included 110 patients (median age 75 years, interquartile range [IQR] 69–80; 82% male); diagnoses were ATTR-CA in 61 and AL-CA in 49. Median fibrosis extent was 10% (IQR 5-15) in the whole cohort (AL-CA, 10% [5-15%]; ATTR-CA, 10% [5-18%]; p=0.640). Fibrosis was focal or plurifocal in both forms (AL-CA: 48% vs 52%; ATTR-CA: 60% vs 40%; p=0.30). Fibrosis did not correlate with total amyloid burden (whole cohort, Spearman rho=−0.122, p=0.230; AL-CA: rho=−0.087, p=0.569; ATTR-CA: rho=−0.160, p=0.251), while it correlated with macrophage and T-cell density (AL-CA: CD68+ rho=0.332, p=0.028; CD3+ rho=0.340, p=0.022; ATTR-CA: CD68+ rho=0.408, p=0.002; CD3+ rho=0.342, p=0.015). In the subset with ECV (n=54; AL-CA n=31, ATTR-CA n=23), ECV tracked amyloid burden more closely than fibrosis (ECV vs amyloid rho=0.442, p=0.001; ECV vs fibrosis rho=0.225, p=0.105), driven primarily by ATTR-CA (ECV vs amyloid rho=0.528, p=0.010; ECV vs fibrosis rho=0.110, p=0.617). In AL-CA, ECV showed only borderline associations with both amyloid and fibrosis (rho=0.327, p=0.083, and rho=0.344, p=0.063, respectively). Conclusions: In patients with LV biopsy–proven CA, fibrosis is common and similar in AL- and ATTR-CA. Fibrosis is more closely related to inflammatory infiltrates than to total amyloid burden, and CMR-derived ECV appears to track amyloid burden more than fibrosis, particularly in ATTR-CA. Together, these results support pursuing adjunct antifibrotic therapies and fibrosis-specific imaging, to better estimate recovery potential and monitor myocardial remodeling after amyloid-removal therapies, rather than relying on ECV alone.