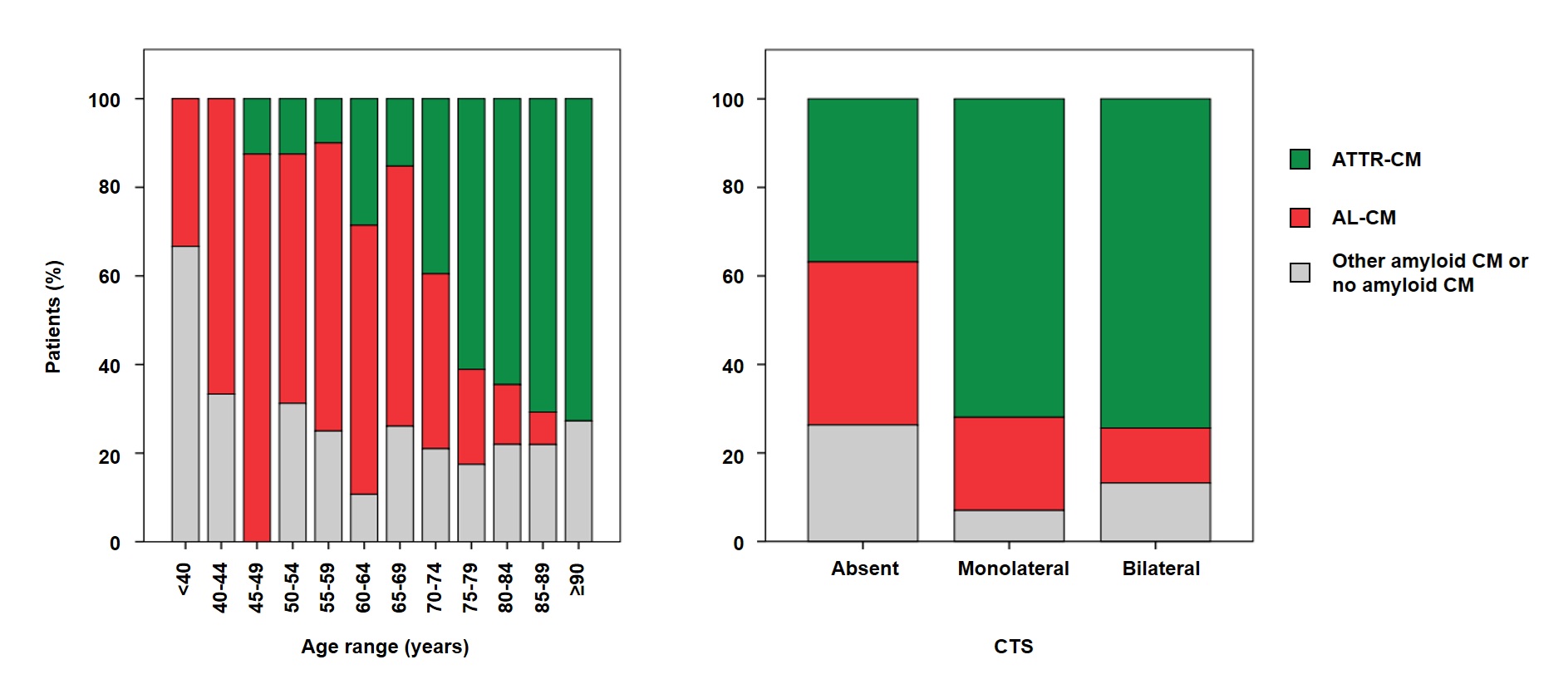
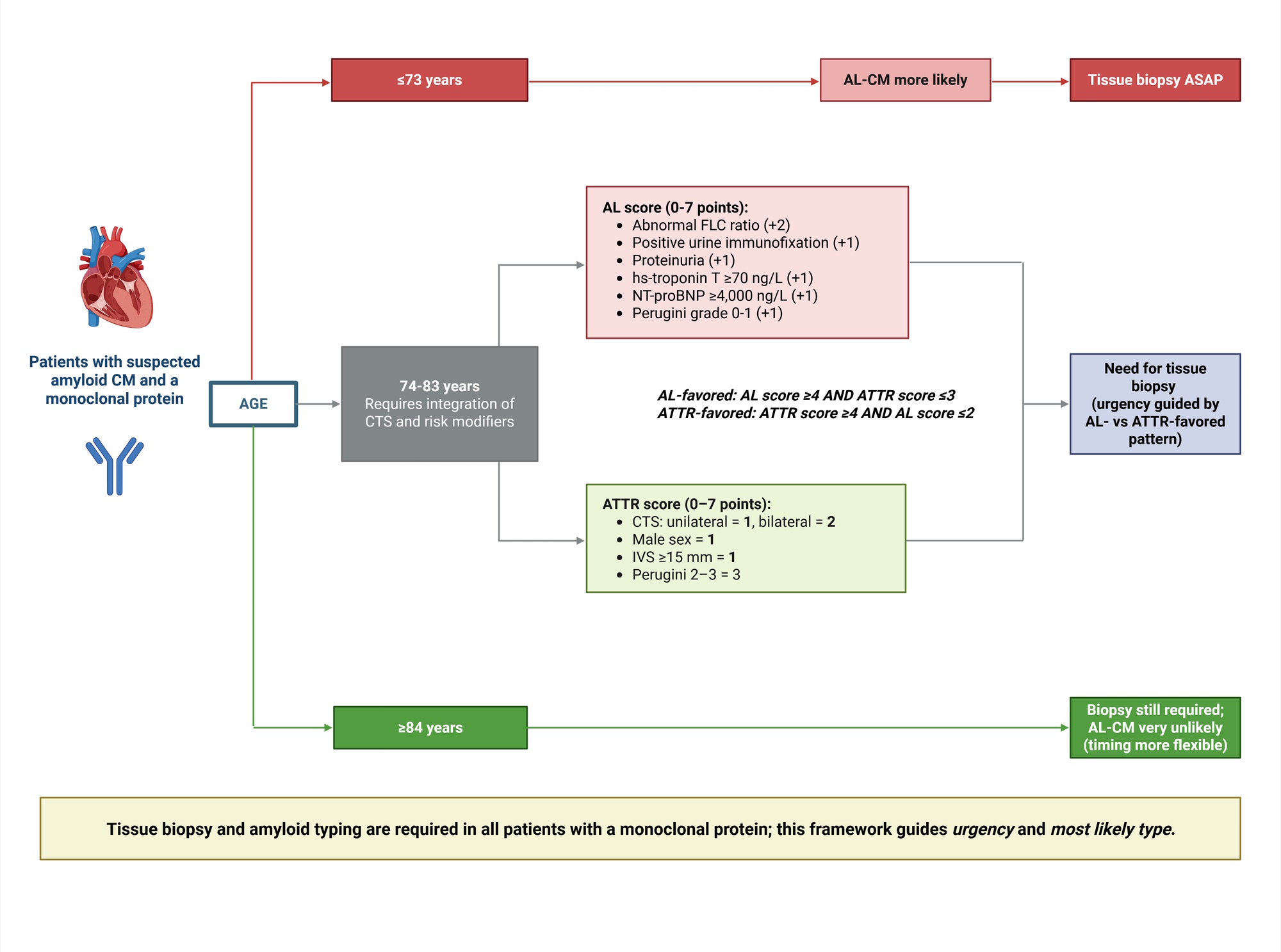
Background: In patients with suspected amyloid cardiomyopathy (CM) and a monoclonal protein, tissue biopsy is mandatory to exclude amyloid light-chain CM (AL-CM), the subtype requiring urgent therapy. We aimed to develop a pragmatic approach to estimate the most likely final diagnosis before biopsy. Methods: We studied a multicenter cohort of adults referred for biopsy because of suspected amyloid CM. Clinical, laboratory, ECG/echocardiographic and bone-tracer scintigraphy data were collected at initial suspicion. In patients with a monoclonal protein, we derived age cut-offs overall and by carpal tunnel syndrome (CTS), defined an age-based “gray zone” (74–83 years), and evaluated risk modifiers for AL-CM and transthyretin CM (ATTR-CM). We built a points-based algorithm for “gray-zone” patients. Clinical utility was assessed using decision-curve analysis. Results: Among 602 patients (median age 76 years; 81% male), 524 (87%) had a monoclonal protein. In the monoclonal-protein subgroup, ATTR-CM accounted for 49%, AL-CM for 30%, and neither ATTR- nor AL-CM for 21%. Age ≤73 years was the Youden-optimal threshold favoring AL-CM (sensitivity 66%, specificity 78%), whereas age >84 years made AL-CM unlikely (sensitivity 98%, negative predictive value 94%), defining a “gray zone” (n=261; ATTR-CM 63%, AL-CM 18%, no amyloid CM 20%). Within the gray zone, abnormal free light-chain ratio, positive urine immunofixation, proteinuria, and higher troponin T and N-terminal pro-B-type natriuretic peptide (NT-proBNP) favored AL-CM, while higher Perugini grade, male sex, greater interventricular septal thickness, and higher NT-proBNP favored ATTR-CM. Penalized models achieved cross-validated area under the curves of 0.87 (AL-CM) and 0.94 (ATTR-CM). A two-score algorithm identified small gray-zone subsets enriched for AL-CM (positive predictive value 81.5%, specificity 98%) or ATTR-CM (positive predictive value 95.2%, specificity 94%). DCA showed higher net benefit for score- and model-based strategies versus treat-all/treat-none across clinically relevant threshold probabilities, with consistent patterns across regions. Conclusions: In patients with suspected amyloid CM and a monoclonal protein, age- and CTS-anchored stratification refined by a compact set of routine laboratory and imaging features provides actionable pre-biopsy probabilities and may help prioritize urgent biopsy and AL-directed work-up while definitive tissue typing is pursued.