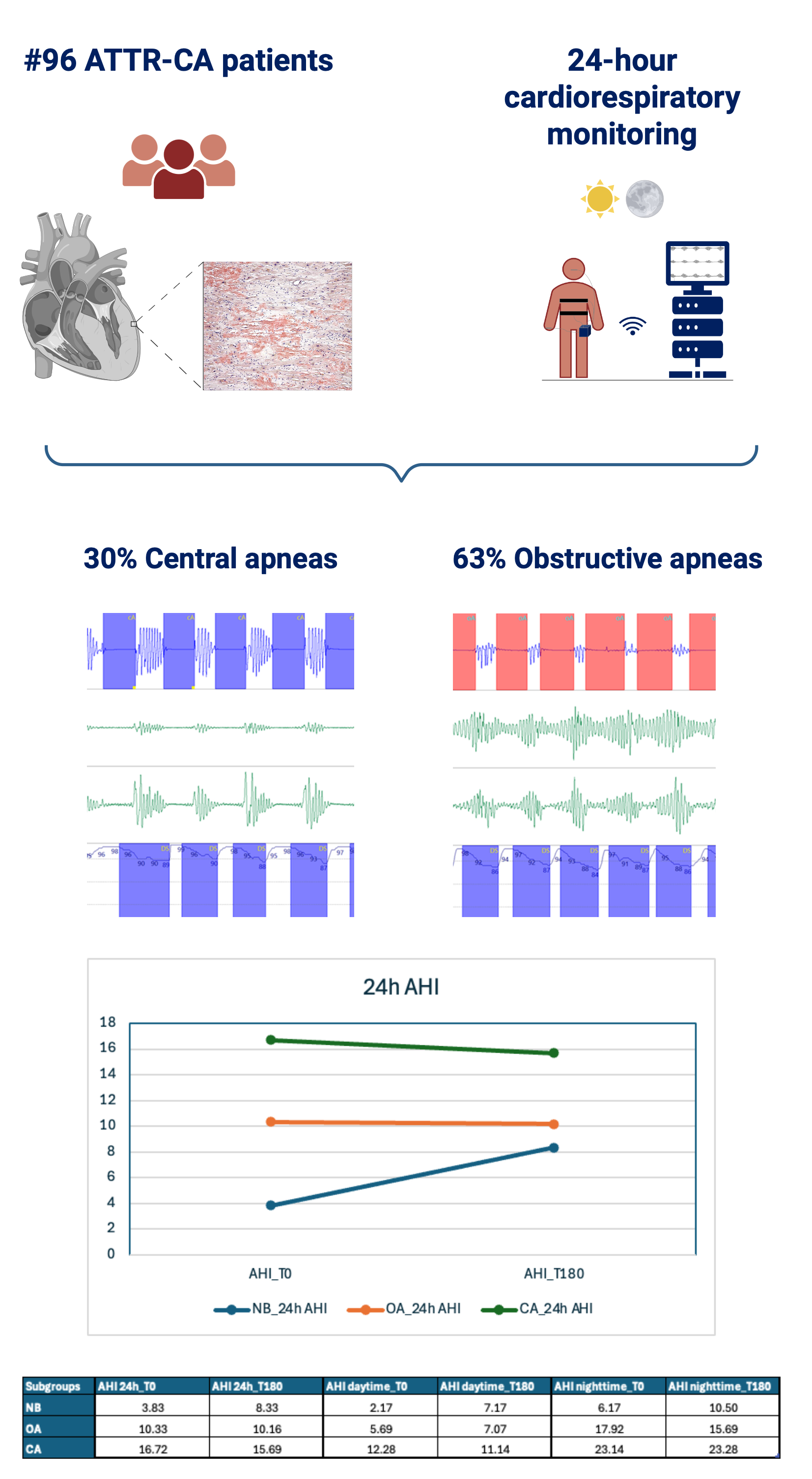
Background: Heart failure (HF) is frequently associated with central apnea (CA) and obstructive apnea (OA), which often improve after treatment. Amyloid transthyretin (TTR) cardiomyopathy (ATTR-CM) is increasingly recognized as a cause of HF. Tafamidis, a TTR stabilizer, slows disease progression by preventing TTR misfolding and aggregation, reducing mortality, cardiovascular hospitalizations, and functional decline. Its potential effect on apnea burden in ATTR-CM remains unclear. Purpose: To evaluate the impact of tafamidis on the apneic burden in patients with ATTR-CM and HF. Methods: Consecutive ATTR-CM patients initiating tafamidis therapy underwent a 24-hour ambulatory cardiorespiratory monitoring to evaluate the prevalence and severity of breathing disorders at baseline and after 6 months. The severity of these disorders was quantified using the apnea-hypopnea index (AHI). Patients were categorized as having CA (AHI >5/hour, ≥50% central), OA (AHI ≥5/hour, >50% obstructive), or normal breathing (NB, AHI <5/hour). Results: Ninety-six ATTR-CM patients (90% males, mean age 78±6 years) were enrolled. At baseline, 63% of patients had OA, and 30% had CA. After 6 months, 66% and 21% of patients presented with OA and CA, respectively. Then after the treatment, AHI remained unchanged over 24 hours (p=0.93), as well as during daytime (p=0.39), and nighttime (p=0.44), regardless of apnea type or baseline severity. When classifying patients according to the type of apneas, tafamidis treatment was not associated with significant changes in daytime, nighttime, or 24-hour AHI (Figure). Conclusions: In a real-life cohort of ATTR-CM patients, 6-month treatment with tafamidis was associated with stabilization of apneic burden without differences between obstructive and central apnea, or between daytime and nighttime events.