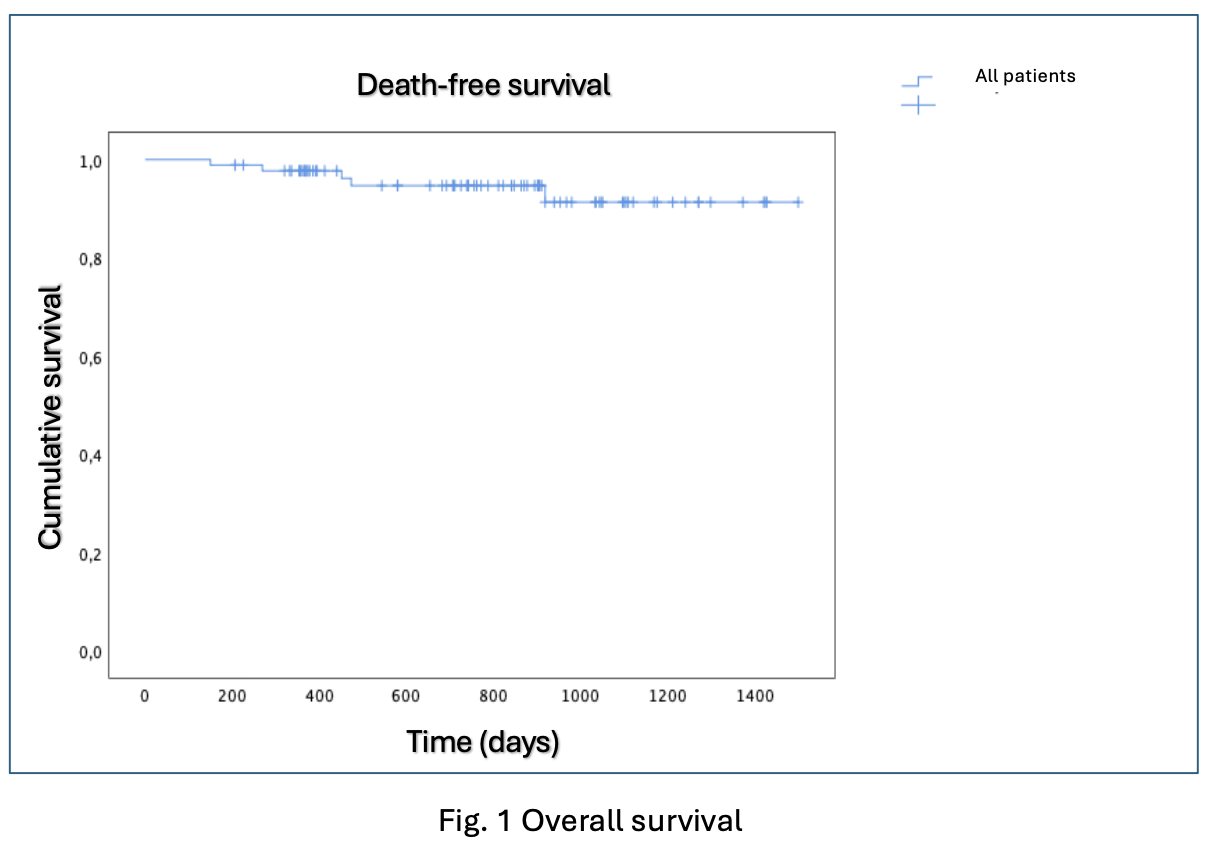
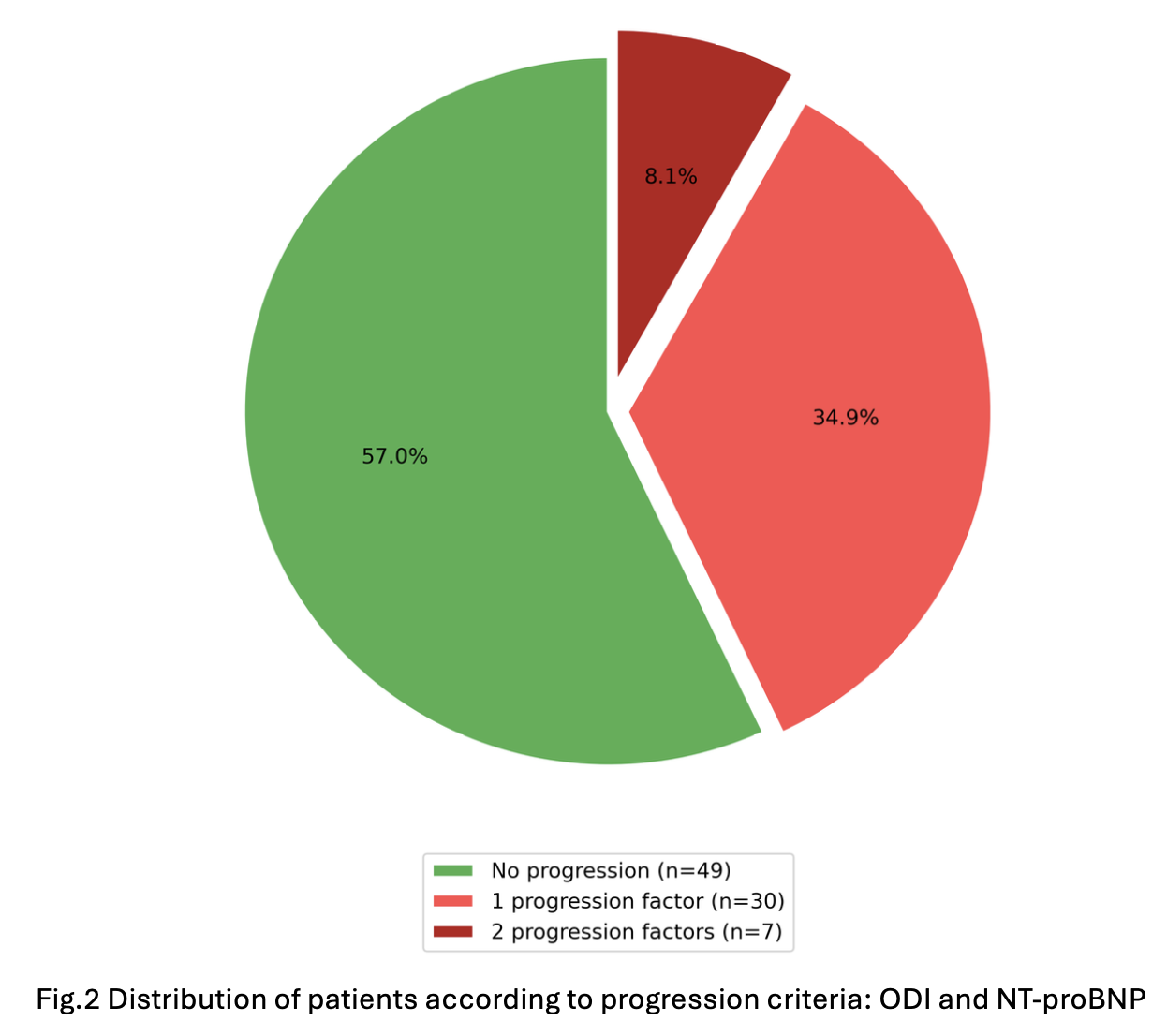
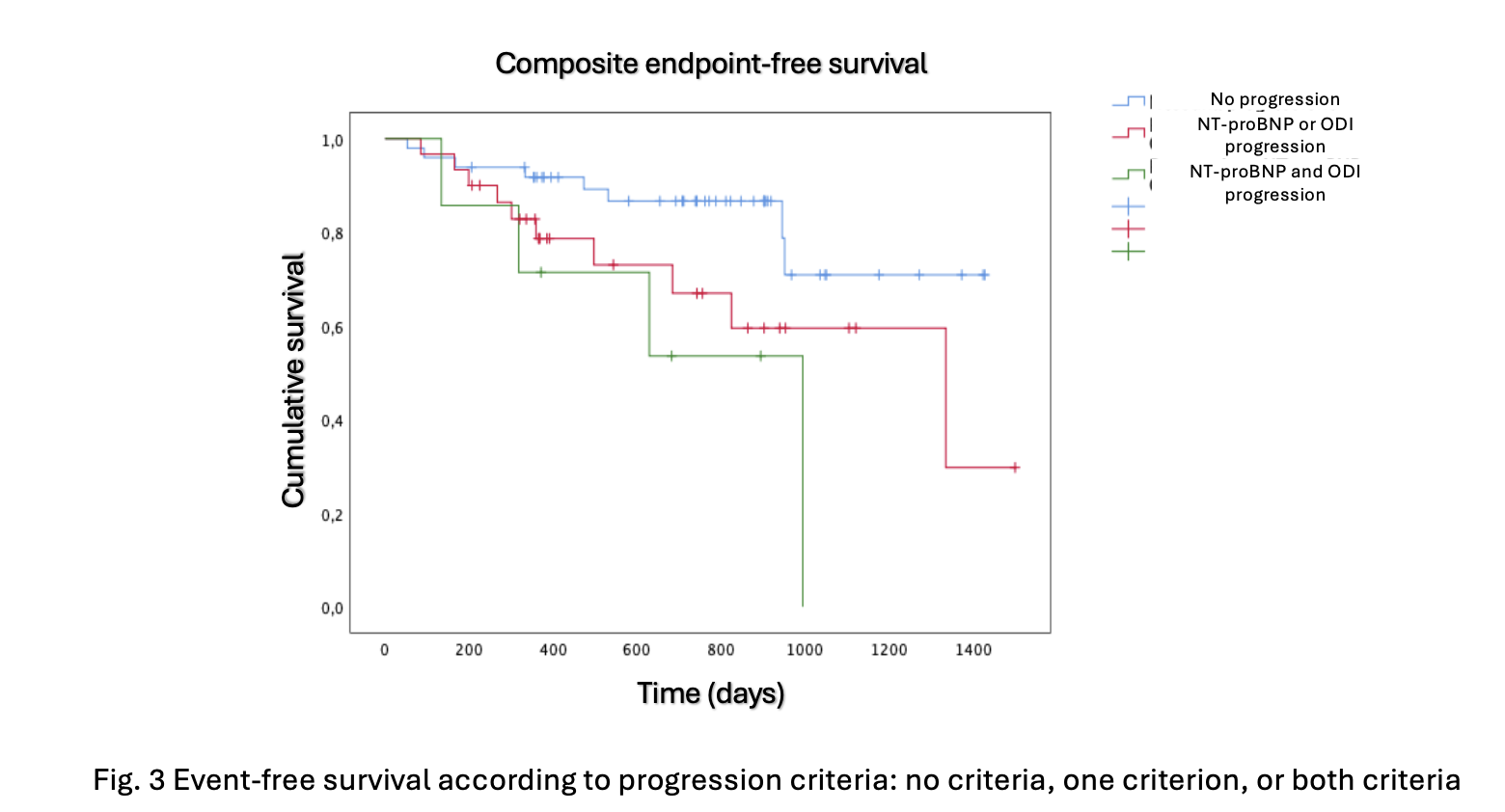
Introduction Tafamidis is the cornerstone therapy for transthyretin amyloid cardiomyopathy (ATTR-CM); however, validated criteria for monitoring disease progression in treated patients are still lacking. Emerging evidence from untreated cohorts suggests that increases in NT-proBNP levels and outpatient diuretic intensification (ODI) may represent markers of progression. Objectives To evaluate all-cause mortality and cardiovascular hospitalizations in a real-world population of NYHA class I–II ATTR-CM patients treated with tafamidis; to assess the frequency of disease progression markers; and to identify those associated with an unfavorable prognosis. Methods This single-center retrospective observational study was conducted at the Cardiomyopathy Unit. Eighty eight consecutive patients with wild-type or variant ATTR-CM, NYHA class I–II, who initiated tafamidis between 2020 and 2024 and had at least 12 months of follow-up were included. The primary endpoint was a composite of all-cause mortality, hospitalization for heart failure requiring iv diuretics, and hospitalization for other cardiovascular causes. Two analyses were performed: a Cox proportional hazards model and a landmark analysis excluding patients with events occurring before the 12-month time point, in which the prognostic value of progression markers between baseline and 12 month, was assessed. After identifying clinically relevant progression criteria, the population was divided into those who experienced progression versus those who didn’t. Results Over a median follow-up period of 26 months, the all-cause mortality rate was 5.7% ( Fig .1 ). The primary endpoint occurred in 28.4% of patients. Thirty patients (34.9%) showed progression defined by at least one marker (NT-proBNP or ODI), while seven patients (8.4%) met both criteria ( Fig.2 ). At multivariable analysis, the presence of a single progression marker was associated with increased risk of the primary endpoint (HR 2.47, 95% CI 0.99–6.27; p=0.05), with a stronger association when both markers were present (HR 4.40, 95% CI 1.32–14.70; p=0.016). ( Fig.3 ) Conclusions: In a contemporary real-world cohort of ATTR-CM patients treated with tafamidis mortality rate was acceptably low. However, a relevant proportion of patients experienced disease progression. NT-proBNP increase and ODI emerged as clinically meaningful markers of progression associated with adverse prognosis, supporting closer follow-up and optimization of therapeutic strategies.